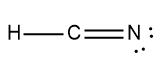11. Bonding & Molecular Structure
Lewis Dot Structures: Acids
11. Bonding & Molecular Structure
Lewis Dot Structures: Acids - Video Tutorials & Practice Problems

Get help from an AI Tutor
Ask a question to get started.
Drawing the Lewis Dot Structure of Acids involves first drawing the polyatomic anion and then attaching the H+ ion.
Lewis Dot Structures of Acids
1
concept
Lewis Dot Structures: Acids
Video duration:
1mPlay a video:
Acids are soluble covalent compounds containing H+ ion.
2
example
Lewis Dot Structures: Acids Example 1
Video duration:
2mPlay a video:
3
Problem
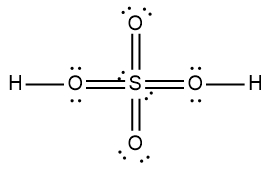
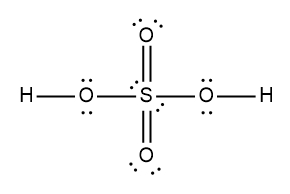
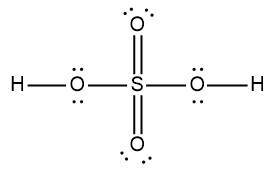
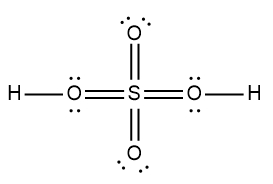
ProblemDetermine the Lewis Dot Structure for the following acid:H2SO4
A
B
C
D
4
Problem
ProblemDetermine the Lewis Dot Structure for hydrocyanic acid:HCN
A
B
C
D
5
Problem
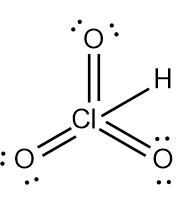
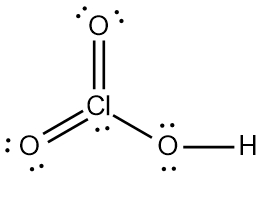
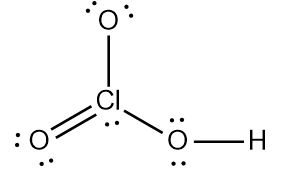
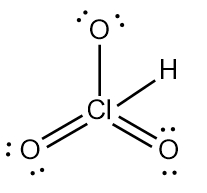
ProblemDetermine the Lewis Dot Structure for chloric acid:HClO3
A
B
C
D
6
Problem
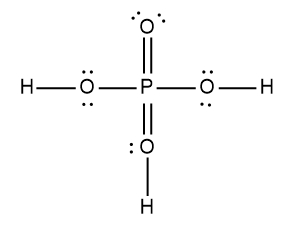
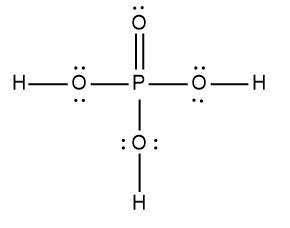
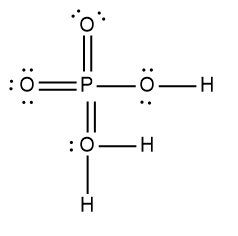
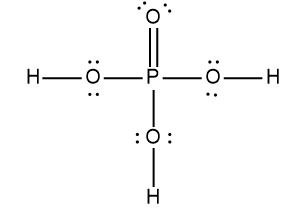
ProblemDetermine the Lewis Dot Structure for phosophoric acid:H3PO4
A
B
C
D
Do you want more practice?
We have more practice problems on Lewis Dot Structures: Acids
Additional resources for Lewis Dot Structures: Acids
PRACTICE PROBLEMS AND ACTIVITIES (9)
- There are many Lewis structures you could draw for sulfuric acid, H2SO4 (each H is bonded to an O). (a) What L...
- White phosphorus reacts spontaneously with the oxygen in air to form P4O6. (c) Which structure obeys the oct...
- White phosphorus reacts spontaneously with the oxygen in air to form P4O6. (b) When P4O6 is dissolved in water...
- Draw the Lewis structure for each compound. c. H3AsO4
- Draw the Lewis structure for each compound. b. H3PO3 (two OH bonds)
- Draw a lewis structure for so2 in which all atoms obey the octet rule. Show formal charges. Do not consider ri...
- Draw a Lewis structure for SO2 in which all atoms obey the octet rule. Show formal charges. Do not consider ri...
- In molecules, C, H, O, and N atoms usually make __, __, __, and __ bonds respectively.
- Determine the number of valence electrons in HCN and then draw the corresponding Lewis structure.