13. Liquids, Solids & Intermolecular Forces
Body Centered Cubic Unit Cell
13. Liquids, Solids & Intermolecular Forces
Body Centered Cubic Unit Cell - Video Tutorials & Practice Problems

Get help from an AI Tutor
Ask a question to get started.
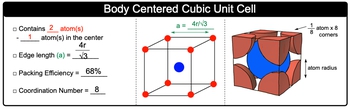
Body Centered Cubic Unit Cell contains 2 atoms total.
Body Centered Cubic Unit Cell
1
concept
Body Centered Cubic Unit Cell Concept 1
Video duration:
1mPlay a video:
2
example
Body Centered Cubic Unit Cell Example 1
Video duration:
40sPlay a video:
3
Problem
ProblemTungsten possesses a body-centered cubic structure. If its density is 19.28 g/cm3, what is its radius in pm?
A
910 pm
B
316 pm
C
137 pm
D
3.18 pm
4
Problem
ProblemVanadium has a body-centered cubic structure. If the atomic radius of vanadium is 134 pm, calculate the density of solid vanadium.
A
5.33 g/cm3
B
4.29 g/cm3
C
5.71 g/cm3
D
0.52 g/cm3
5
Problem
ProblemThe edge of a body-centered cubic unit cell of an element Z was found to be 2.88 x 10-8 cm. The density of the element is 7.2 g/cm3. What is the approximate molar mass of Z?
A
101.7 g•mol-1
B
39.107 g•mol-1
C
10.291 g•mol-1
D
51.996 g•mol-1
Do you want more practice?
We have more practice problems on Body Centered Cubic Unit Cell
Additional resources for Body Centered Cubic Unit Cell
PRACTICE PROBLEMS AND ACTIVITIES (20)
- Identify each of the following kinds of packing: (d)
- Determine the number of atoms per unit cell for each metal. (b) Tungsten
- Sodium metal (atomic weight 22.99 g>mol) adopts a body-centered cubic structure with a density of 0.97 g>...
- Molybdenum crystallizes with the body-centered unit cell. The radius of a molybdenum atom is 136 pm. Calculate...
- Calcium crystallizes in a body-centered cubic structure at 467°C. (a) How many Ca atoms are contained in each...
- Barium has a density of 3.59 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radi...
- Calcium crystallizes in a face-centered cubic unit cell at room temperature that has an edge length of 5.588 ...
- Tungsten crystallizes in a body-centered cubic unit cell with an edge length of 317 pm. What is the length in...
- Sodium has a density of 0.971 g>cm3 and crystallizes with a body-centered cubic unit cell. What is the rad...
- If a protein can be induced to crystallize, its molecular structure can be determined by X-ray crystallograph...
- Iron crystallizes in a body-centered cubic unit cell with an edge length of 287 pm. Iron metal has a density ...
- Cesium chloride crystallizes in a cubic unit cell with Cl- ions at the corners and a Cs+ ion in the center. C...
- Potassium metal crystallizes in a body-centered cubic structure. Draw one unit cell, and try to draw an elect...
- The unit cell of a compound containing Co and O has a unit cell shown below. The Co atoms are on the corners, ...
- An unknown metal is found to have a density of 7.8748 g/cm3 and to crystallize in a body-centered cubic lattic...
- What is the coordination environment of the K+ ions in the fullerene-based superconductor K3C60?
- A cube-shaped crystal of an alkali metal, 1.62 mm on an edge, was vaporized in a 500.0 mL evacuated flask. Th...
- A cube-shaped crystal of an alkali metal, 1.62 mm on an edge, was vaporized in a 500.0 mL evacuated flask. The...
- Vanadium atom has a radius of 131 pm and crystallizes with a bcc unit cell. Determine the number of unit cells...
- Titanium metal (d = 4.50 g/cm3) has a body-centered cubic unit cell. Calculate the edge length of the unit cel...

